You are here
Home 🌿 Cannabis Technology News 🌿 Succesful feasibility study for the development of topical medical cannabis products 🌿Succesful feasibility study for the development of topical medical cannabis products
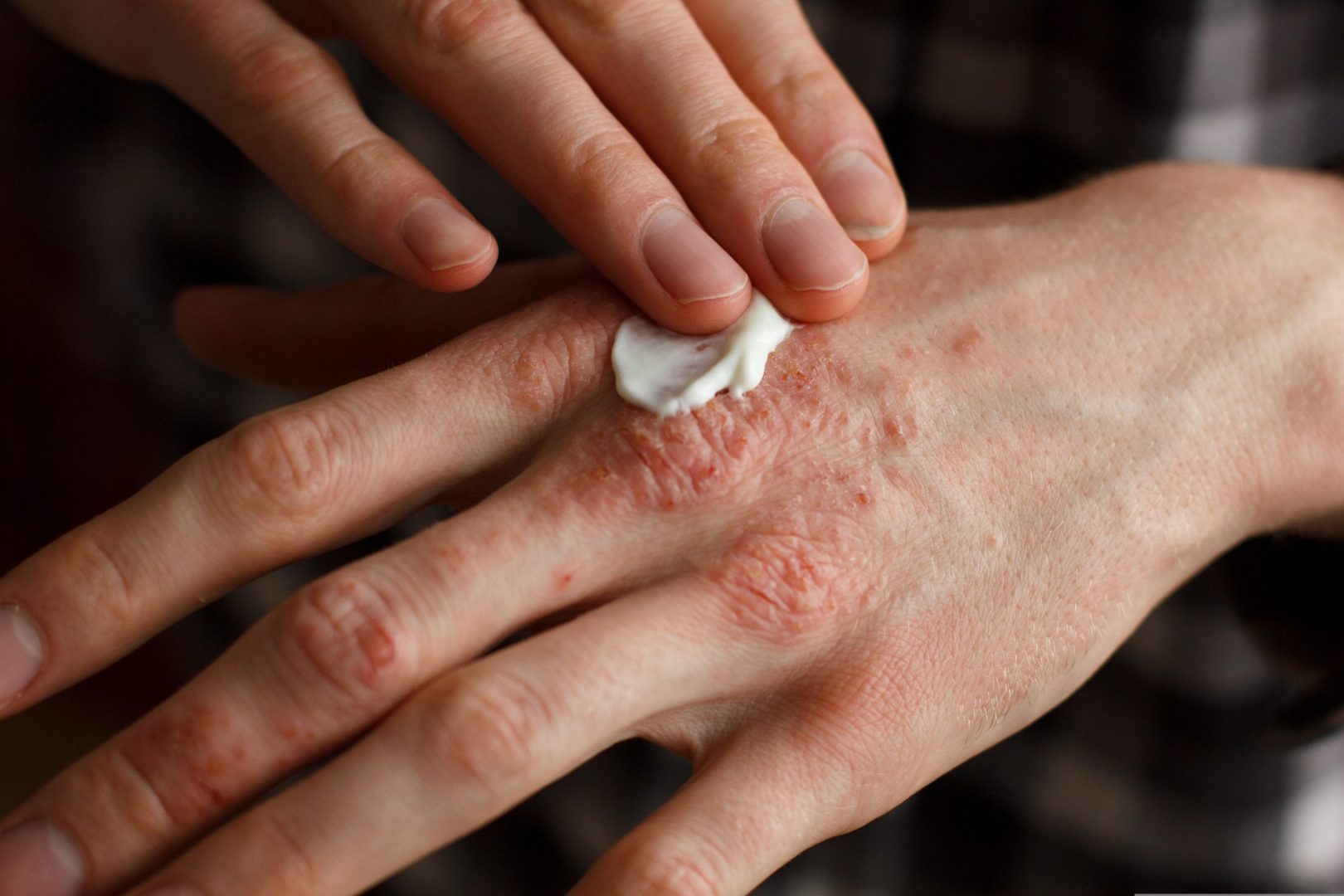
Cannassure Therapeutics Ltd (TASE: CSURE) (“Cannassure”), an Israel based developer and producer of innovative medical cannabis products, announces today the successful completion of a feasibility study for the development of IP protected, homogeneous, topical medical Cannabis products for the treatment of skin inflammation, including psoriatic lesions. (PRESS RELEASE)
Product development of such topical medical Cannabis products based on AKVANO® is being conducted under a collaboration between Cannassure and Lipidor AB (“Lipidor”), a Swedish topical drug development company, owner of the proprietary drug delivery platform technology AKVANO™, which was signed in December 2019. The development project budget is partially funded by the Israeli Innovation Authority, by a non-dilutive grant.
The product under development is an incorporation of cannabinoids with AKVANO®, a topical drug delivery platform. Successful development of the product will allow localized administration of a precise dosage of cannabinoids using a spray, in a technology that allows for prolonged contact and optimal penetration of the active ingredient into the skin.
As part of the feasibility study, AKVANO® technology and cannabinoids were tested for compatibility, and it was found that the cannabinoids can be successfully combined in the AKVANO® system for a homogeneous, uniform and stable formulation. Cannassure tested the degree of product penetration into the skin by using known and acceptable models and found significant penetration of active ingredients into the skin tissue. This enhanced penetration may increase the therapeutic potential of the product. Based on these results, Cannassure will proceed to accelerated development stages and more advanced testings.
According to the agreement, Cannassure has informed Lipidor on its intention to start negotiating an exclusive global license for the use of Lipidor’s technology for the development of cannabinoids based topical therapeutics products for the treatment of indications to be chosen by Cannassure.
The main guidelines for the licencing agreement, including royalties to be paid to Lipidor on product sales, were defined in the collaboration agreement.
According to estimates, the global market for products for the treatment of psoriasis is expected to reach 21.1 billion dollars by 2022. The current global market for topical products for the treatment of psoriasis is valued at 7 billion dollars, and the global market for topical pain treatment products is projected to reach 13 billion dollars.

Cannassure Growing Facility (Credit: Cannassure PR)
The product development of topical treatments based on cannabinoids is in line with Cannassure’s strategy focused on the development and commercialisation of innovative medical cannabis products based on established drug delivery systems and formulations for enhanced therapeutic value. The product development is performed under the Israeli Medical Cannabis regulation, which allows a short development process.
About Cannassure Therapeutics Ltd.
Cannassure Therapeutics Ltd. (TASE:CSURE) is a leading, world class, trusted developer and provider of top-quality-grade medical cannabis products and pharmaceutical cannabinoid medicines, addressing a broad range of unmet medical needs.
About Lipidor AB
Lipidor AB (www.lipidor.se) is a Swedish, Stockholm-based research and development company with a pipeline of pharmaceutical development projects in preclinical and clinical phases. The Company develops topical medical products for the treatment of diseases such as psoriasis, bacterial skin infections and atopic dermatitis by reformulation of proven pharmaceutical substances.
420 Intel is Your Source for Marijuana News
420 Intel Canada is your leading news source for the Canadian cannabis industry. Get the latest updates on Canadian cannabis stocks and developments on how Canada continues to be a major player in the worldwide recreational and medical cannabis industry.
420 Intel Canada is the Canadian Industry news outlet that will keep you updated on how these Canadian developments in recreational and medical marijuana will impact the country and the world. Our commitment is to bring you the most important cannabis news stories from across Canada every day of the week.
Marijuana industry news is a constant endeavor with new developments each day. For marijuana news across the True North, 420 Intel Canada promises to bring you quality, Canadian, cannabis industry news.
You can get 420 Intel news delivered directly to your inbox by signing up for our daily marijuana news, ensuring you’re always kept up to date on the ever-changing cannabis industry. To stay even better informed about marijuana legalization news follow us on Twitter, Facebook and LinkedIn.




